So remember my last Research blog update where I talked about the Perry Lab endeavors to find the best possible way to smush lice for DNA extractions? Well I'm at it again!
As I've previously mentioned, there are three main tasks to complete when beginning a new DNA sequencing project: 1) get the DNA out of your sample, 2) prepare your DNA libraries for sequencing, and 3) do both 1 and 2 in a cost-effective manner. I dedicated yesterday to step 2 of this process.
| A DNA library is pretty much what it sounds like: all of the genomic DNA from an individual sorted and organized in such a way that it is more amenable to sequencing. A good portion of genomic sequencing is performed on machines made by Illumina, so naturally they've also created kits for preparing your DNA libraries. The Perry Lab has had success with these kits, but recall point 3 from above: costs. Kits are generally more expensive than what are referred to as "in-house" protocols because you can typically buy the same reagents you would use in the kit in bulk for a lower price. Enter Matthias Meyer and Martin Kircher. |
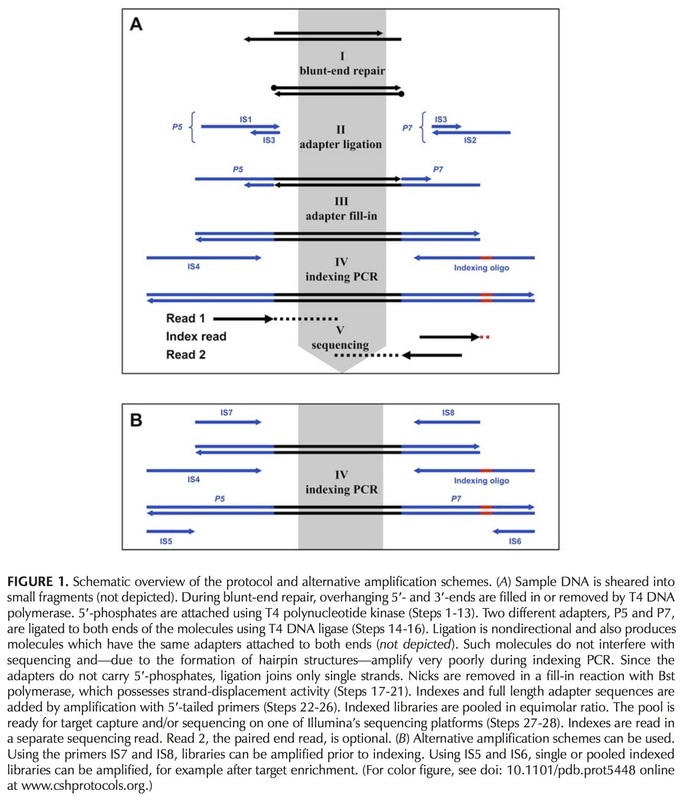
Meyer & Kircher's protocol Illumina sequencing library preparation for highly multiplexed target capture and sequencing (M&K) has become a Perry Lab standard. M&K is nearly identical to Illumina's equivalent library prep protocol (see above figure), with the only real difference being the making of master mixes. The nice thing about kits is that every reagent you'll need is already prepared for you in a proprietary concoction, so you can just grab and go with each step. For M&K, you have to put together the same ingredients that are found in the ready-made mixes. This adds a bit of time, but in the end the procedure is a bit more controlled by the research group, which is handy if protocols need to be adapted for any particular reason (ours is ancient DNA).
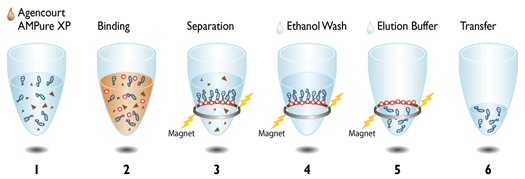
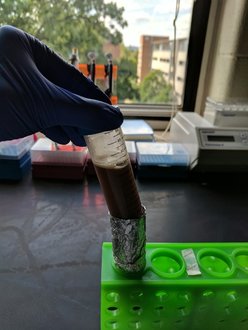
| The really cool thing about both of these protocols is the use of magnetic beads to directly manipulate the DNA strands in their solution (see depiction at left for Agencourt AMpure XP beads). We make our own Solid Phase Reversible Immobilization (SPRI) beads in-house for use in both our ancient and modern lab spaces. The sort of gross looking tube of brown to the left contains these teeny tiny SPRI beads that we can use to "clean" our DNA throughout the library prep process. We can also manipulate the ratio of beads to DNA to select DNA fragments of whatever length we are targeting - in DNA sequencing, length matters. (For those who are curious, the tube is wrapped in foil because the beads are photosensitive and will degrade if exposed to too much light.) |




 RSS Feed
RSS Feed
